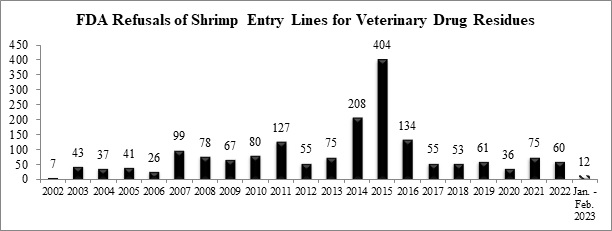
Over the weekend, the U.S. Food and Drug Administration (FDA) has published detailed data regarding 48 seafood entry line refusals in February, of which two (4.2%) were of shrimp for reasons related to banned antibiotics. In addition, the FDA has also published information regarding another 31 seafood entry line refusals in January, of which one (3.2%) was of shrimp for veterinary drug residues. Beyond these refusals, the FDA further released information regarding the refusal of another nine entry lines of shrimp – eight of which were from India – contaminated with salmonella and/or filth.
Through the first two months of this year, the FDA has refused a total of twelve entry lines of shrimp for banned antibiotics, ahead of the pace of total refusals last year.
The three entry lines recently reported of shrimp refused for antibiotic residues in January and February were for shipments from the following two shrimp exporters:
- Mangala Seafoods (India), a company that was added to Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) for its shipments of shrimp on March 1, 2023 and was added to Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”) for chloramphenicol in its shrimp on January 25, 2023, had one entry line refused for shrimp contaminated with veterinary drug residues by the Division of Southwest Imports on January 30, 2023 and one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Northern Border Imports on February 1, 2023; and
- Hoang Phong Seafood Co. (Vietnam), a company that is not currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”), Import Alert 16-127 (“Detention Without Physical Examination of Crustaceans Due to Chloramphenicol”), or Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”), had one entry line refused for shrimp contaminated with nitrofurans by the Division of Southeast Imports on February 17, 2023.
In addition, the FDA’s updated reporting indicated that the agency refused another five entry lines of shrimp for the presence of salmonella from an exporter in Indonesia (PT. Indokom Samudra Persada; January 30, 2023; Division of Northeast Imports) and from four different exporters in India: Kader Exports Private Limited (January 23, 2023; Division of Northeast Imports); Edhayam Frozen Foods Pvt Ltd. (January 17, 2023; Division of West Coast Imports); HT Foods Pvt Ltd (January 26, 2023, Division of Northeast Imports); and Royale Marine Impex Pvt. Ltd. (February 16, 2023; Division of West Coast Imports).
The FDA also reported that another four entry lines of Indian shrimp were refused for filth: Kader Exports Private Limited (February 22, 2023; Division of Northeast Imports); Sandhya Aqua Exports Pvt Ltd (February 24, 2023; Division of West Coast Imports); Royale Marine Impex Pvt. Ltd. (February 10, 2023; Division of West Coast Imports); and Highland Agro (January 27, 2023; Division of Northeast Imports).
