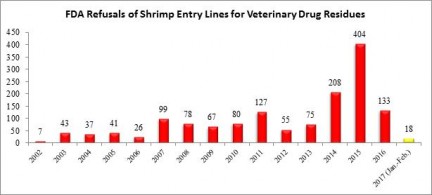
Over the weekend, the U.S. Food and Drug Administration’s (FDA) released information regarding entry line refusals for February. In total, 10 of the 122 (8.2%) of the entry line refusals reported for last month were of shrimp for reasons related to banned antibiotics.
The ten shrimp entry lines refused by the FDA for banned antibiotics in February were from China, Hong Kong, Malaysia and Vietnam and were reported by two different FDA districts:
- Minh Phu Seafood Corporation (Vietnam), a company that was added to Import Alert 16-124 for sulfamethoxazole in its shrimp on February 22, 2017 after its affiliate Minh Qui Seafood Co., Ltd. had been added to that Import Alert for sulfamethoxazole in its shrimp on July 22, 2016, had one entry line refused for shrimp contaminated with veterinary drug residues in the New York District;
- Suntraco Food Industries Sdn. Bhd. (Malaysia), a company located in Peninsular Malaysia that has not been exempted from Import Alert 16-136, had one entry line refused for shrimp contaminated with veterinary drug residues in the New York District;
- Lee Fung Marine Products Trading Co. (Hong Kong), a company that is not currently listed on Import Alert 16-124, Import Alert 16-127, or Import Alert 16-129, had two entry lines refused for shrimp contaminated with veterinary drug residues and unsafe additives in the New York District;
- Yantai Fujiyasu Food Company Limited (China), a company that has not been exempted from Import Alert 16-131, had one entry line refused for shrimp contaminated with veterinary drug residues and unsafe additives in the New York District;
- Wuhan City Yingnadi Int’l Trade (China), a company that has not been exempted from Import Alert 16-131, had one entry line refused for shrimp contaminated with veterinary drug residues and unsafe additives in the San Francisco District;
- Dalian Shanhai Seafood Co., Ltd. (China), a company that has not been exempted from Import Alert 16-131, had two entry lines refused for breaded shrimp contaminated with veterinary drug residues and unsafe additives in the New York District; and
- Ruian Huasheng Aquatic Products (China), a company that has not been exempted from Import Alert 16-131, had two entry lines refused for shrimp contaminated with veterinary drug residues and unsafe additives in the New York District.
There have not been reported refusals of entry lines for reasons related to antibiotics for shrimp shipped from Hong Kong since April of 2014. Before last month, the FDA’s public records going back to 2002 indicate that there had only been a total of eleven entry line refusals of shrimp shipped from Hong Kong for reasons related to banned antibiotics. The six entry lines of shrimp shipped from China, from four different companies, was the most refused in a month since March of 2016.
The FDA’s reporting may indicate that in response to the Import Alert on shrimp shipped to the United States from peninsular Malaysia, Chinese shrimp producers and exporters are exploring other ways to enter the U.S. market. These shipments continue to raise concerns regarding the use of banned antibiotics in shrimp aquaculture in China, the world’s largest producer of farmed shrimp.