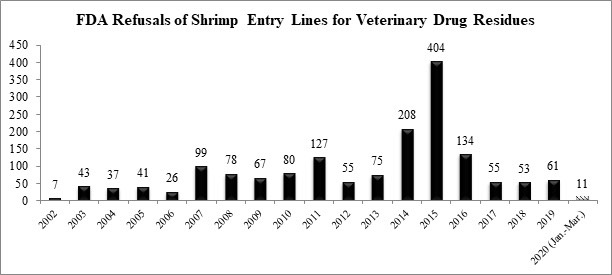
The U.S. Food and Drug Administration (FDA) published data over the weekend reporting that there were 106 total seafood entry line refusals in March, of which 7 were of shrimp for reasons related to banned antibiotics (6.6%).
The FDA has now reported a total of eleven refusals of shrimp entry lines for reasons related to banned antibiotics in 2020.
The seven shrimp entry lines refused in March were for shipments from four different companies in three separate countries, with five of the seven originating in Mexico:
- Negocio Agricola San Enrique S.A. de C.V. (Mexico), a company that is currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”) as of January 7, 2020 for ciprofloxacin and enrofloxacin in its shrimp, had four entry lines refused for shrimp contaminated with veterinary drug residues by the Division of Southwest Imports on March 23, 2020;
- Maroa Importaciones (Mexico), a company that is not currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”), Import Alert 16-127 (“Detention Without Physical Examination of Crustaceans Due to Chloramphenicol”), and Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”), had one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Southwest Imports on March 20, 2020;
- Ram’s Assorted Cold Storage Limited (India), a company that is currently listed on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of October 10, 2019, had one entry line refused for shrimp contaminated with nitrofurans by the Division of Northern Border Imports on March 31, 2020; and
- Fuqing Yihua Aquatic Food Co., Ltd. (China), a company that is no longer green-listed on Import Alert 16-131 (“Detention Without Physical Examination of Aquacultured, Shrimp, Dace, and Eel from China – Presence of New Animal Drugs and/or Unsafe Food Additives”), had one entry line refused for breaded shrimp contaminated with veterinary drug residues and an unsafe additive by the Division of Southwest Imports on March 13, 2020.
